Mar 11, 2026
Headline Patrol: Stupp’s Supramolecules, Dancing In a Lab Dish
Sam Maddox
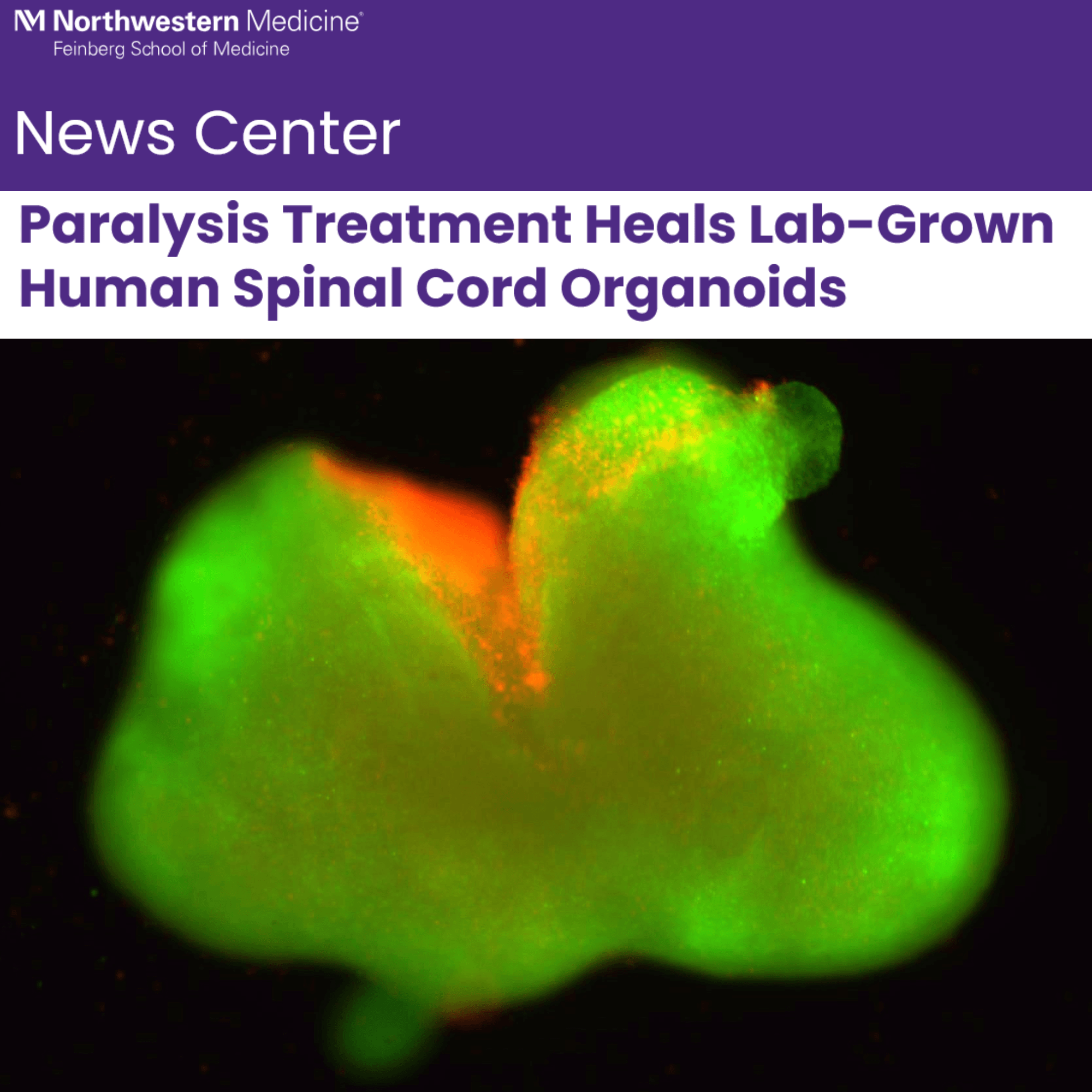
This recent headline - Paralysis Treatment Heals Lab-Grown Human Spinal Cord Organoids - is from the PR department at Northwestern. It describes a spinal cord-in-a-dish preparation that responds to a molecular engineering manipulation. Judge for yourself whether this crosses the threshold of editorial overreach.
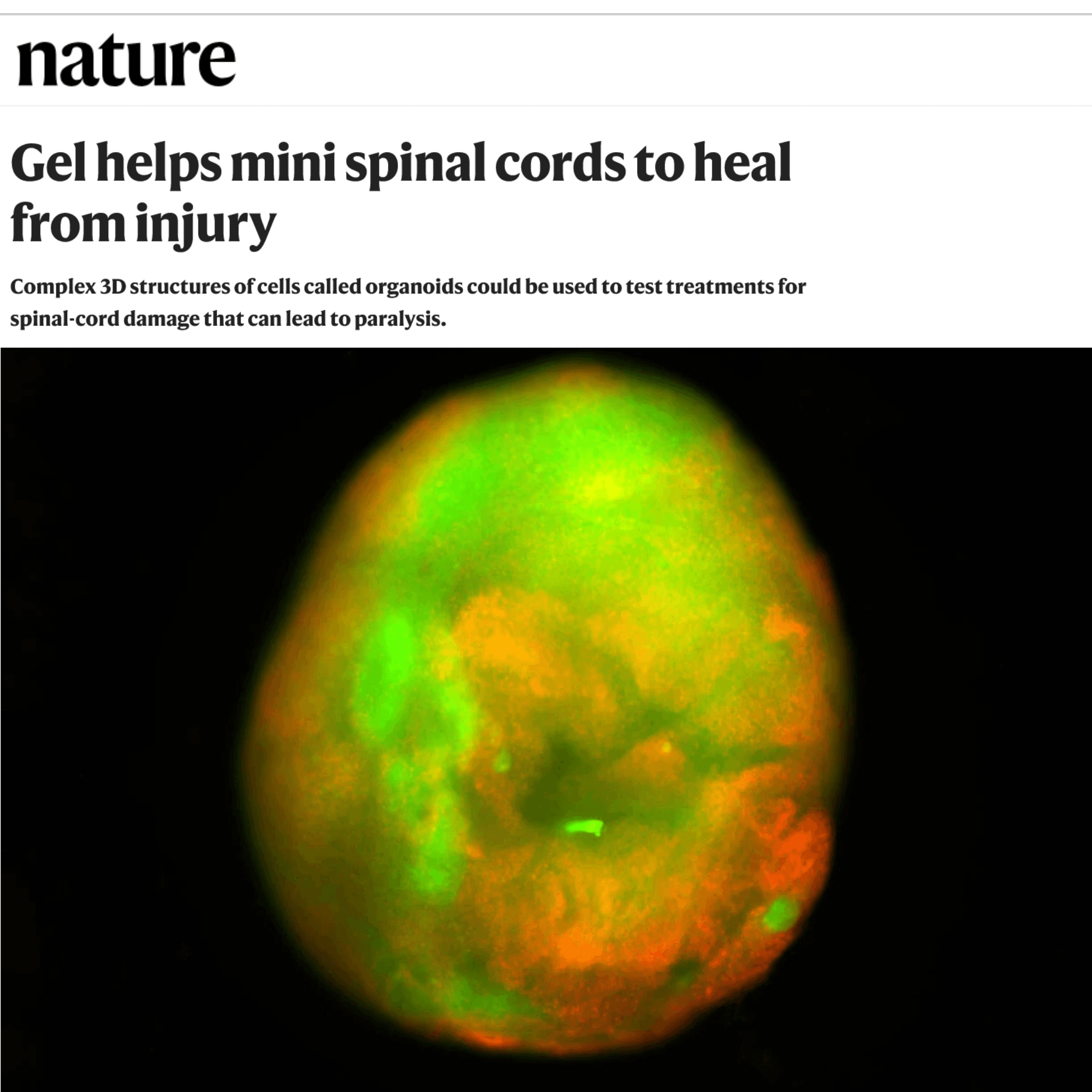
The study, based on a paper titled “Gel helps mini spinal cords to heal from injury” extends our coverage of spinal cord scaffolding (we recently discussed 3-D printed organoids that improved injured animals). This new one may be familiar: it’s a “dancing molecules” update, from the Samuel Stupp lab at Northwestern. In this case he’s using his tiny nanostructures loaded with growth peptides to repair a cell culture in a lab dish.
We’ve previously noted (in a Headline Patrol, on an episode of CureCast, and at our 2022 Symposium) that Stupp’s molecules had a positive effect in acutely injured animals: a one-time injection of supramolecular therapeutic peptides along with the nano scaffold applied 24 hours after severe injury helped mice regain the ability to walk in four weeks.
Here’s the CliffsNotes on the molecular cha cha from Northwestern, where Stupp is a professor of Materials Science & Engineering, Chemistry, Medicine, and Biomedical Engineering:
Injected as a liquid, the dancing molecules therapy immediately gels into a complex network of nanofibers that mimic the extracellular matrix of the spinal cord. By fine-tuning the collective motion, or “dancing,” of the molecules within the nanofibers, Stupp’s team found the therapy connects more effectively with constantly moving cellular receptors.
“Given that cells themselves and their receptors are in constant motion, you can imagine that molecules moving more rapidly would encounter these receptors more often,” Stupp said in 2021. “If the molecules are sluggish and not as ‘social,’ they may never come into contact with the cells.”
From Dish to Animal and Back to Dish?
Normally, positive animal data moves research to the right side of the ledger, toward translation and human application. In this study, the Stupp team steps off an allemande left. Lab dish organoid modeling is very common these days, for every sort of brain disorder and organ system, and the models are so much better now that some say dish models may hasten the decline of animal studies. (Sidenote: animal experiments may be on the way out. Read “The age of animal experiments is waning. Where will science go next?” in Nature for more context.) The Stupp study validates the previous animal work but it doesn’t move closer to clinical relevancy, that is, toward a bigger animal model or a human trial.
Instead, Stupp’s group created spinal cord organoids from a cellular soup derived from human stem cells, including many of the ingredients found in a real mammalian spinal cord, and following the developmental recipe when the nervous system is first formed.
From Northwestern:
Grown in the lab from induced pluripotent stem cells, organoids are miniature, simplified versions of human organs. Although they are only partial organs, organoids mimic the tissue structure, cellular complexity and function of the real thing. This sophisticated mimicry makes organoids ideal for modeling human diseases, testing therapeutics and understanding organ development. Compared to testing treatments in animals and humans, testing in organoids is faster and much less expensive.
Stupp’s team grew the spinal cord organoids from stem cells over the course of months, allowing them to develop complex features including neurons and astrocytes. The team also was the first to add microglia — immune cells in the central nervous system — to simulate inflammatory responses to traumatic spinal cord injury.
“It’s kind of a pseudo-organ,” Stupp said. “We were the first to introduce microglia into a human spinal cord organoid, so that was a huge accomplishment. It means that our organoid has all the chemicals that the resident immune system produces in response to an injury. That makes it a more realistic, accurate model of spinal cord injury.”
Stupp “injured” the little organoids (a few millimeters long) in two ways, by slicing them and by crushing them. Using biological markers, it was obvious the organoid was damaged. Then Stupp’s lab dosed them with his dancing molecule mix — a nano matrix loaded with growth factors. The organoids responded with some nerve fiber growth and measurable reduction of scar formation.

You can listen back to Matthew and Jason’s original interview with Dr. Stupp in episode 60 of our podcast by clicking on the image.
From the Northwestern press release:
When applied to the injured organoids, the liquid therapy gelled to form a scaffold. The therapy calmed inflammation, reduced glial scarring, caused neurites to extend and encouraged neurons to grow in neat, organized patterns.
The scientists demonstrated that human spinal cord organoids can accurately mimic the key effects of spinal cord injury, including cell death, inflammation and glial scarring, a dense mass of scar tissue that creates a physical and chemical barrier to nerve regeneration.
Stupp, from the release: "After applying our therapy, the glial scar faded significantly to become barely detectable, and we saw neurites [nerve fiber extensions] growing, resembling the axon regeneration we saw in animals. This is validation that our therapy has a good chance of working in humans.”
What about chronic SCI? Stupp’s team says it will build even more advanced organoids, including one that models older, chronic injuries that usually have more scar tissue.
Maybe as important as the organoid story is the company founded on Stupp’s research, Amphix Bio. The company’s nano-peptide product AMFX-200 has secured an Orphan Drug Designation for acute SCI from the FDA, which could fast-track translation. Amphix also just got a research grant from the Christopher & Dana Reeve Foundation and U.K.-based Spinal Research to test the combination of AMFX-200 with structured physical rehabilitation in acute and chronic SCI models.
There will certainly be more coming from this company, and from Stupp’s lab. We’ll cover it as it develops.
Stay Curious.
PS - Many of U2FP’s followers have inquired about a Brazilian treatment that is back in the news. This is the one using a polylaminin injection, which I reported on here last year. In a study led by Tatiana Coelho de Sampaio, a scientist at the Federal University of Rio de Janeiro (UFRJ), eight patients with acute SCI got an injection of polylaminin. There was no documentation published but the experimental data was reported by Coelho de Sampaio: Six converted to AIS C, a meaningful level of recovery. Three died, due to pneumonia and sepsis, a meaningful level of uncertainty about the treatment.
The news here is that Brazilian regulatory authorities (Anvisa) have given this study a go-ahead to conduct a small safety trial enrolling five thoracic SCI patients within three days of injury. The trial is sponsored by Cristália Produtos Químicos Farmacêuticos Ltda. More reporting to come, once we get some data.
2026 Science & Advocacy Symposium Sponsors